Leptospirosis
Queensland Health Guidelines for Public Health Units
Revision History
| Version | Date | Changes |
|---|---|---|
| 1.0 | December 2010 | Full revision of guideline. |
| 1.1 | January 2011 | Change to laboratory suggestive evidence and reporting of probable cases. |
| 2.0 | September 2014 | Full revision of guideline (published January 2015) |
| 2.1 | August 2017 | Change to notification process |
| 2.2 | May 2024 | Change to notification process, inclusion of workplace outbreak criteria and minor revision |
Infectious Agent
Leptospirosis is caused by infection with Leptospira species which are helical Gram-negative aerobic bacteria. There are at least 18 species, eight of which are classified as ‘pathogenic’. Each species is further subdivided into serovars. (1)
Case Definitions and Notification Criteria
Report both confirmed and probable cases.(2)
Confirmed case
A confirmed case requires laboratory definitive evidence only.
Laboratory definitive evidence
Isolation of pathogenic Leptospira species
OR
A fourfold or greater rise in Leptospira microscopic agglutination titre (MAT) between acute and convalescent phase sera obtained at least two weeks apart and preferably conducted at the same laboratory
OR
A single Leptospira MAT greater than or equal to 400 supported by a positive enzyme-linked immunosorbent assay IgM result.
Probable case
A probable case requires laboratory suggestive evidence only.
Laboratory suggestive evidence
Detection of pathogenic Leptospira species from a clinical specimen by nucleic acid testing.
Community outbreak criteria
Two or more epidemiologically linked confirmed cases with a suspected local source of exposure.
Workplace outbreak criteria
Two or more cases within a three-month period in a workplace.
Notification Procedure
Pathology Laboratories
To notify on pathological confirmation by usual means.
Lab Aspects
Appropriate specimen collection for suspected leptospirosis cases should be informed by the timeline below, and should include:
For suspected leptospirosis cases presenting less than 7 days from symptom onset:
- At presentation:1 collect blood for culture2 and serum for PCR
- Once 7 days from onset have elapsed: collect serum for enzyme immunoassay (EIA) IgM and MAT
- At least 14 days after acute MAT collection: collect serum for convalescent MAT3
For suspected leptospirosis cases presenting between 7 and 10 days from symptom onset:
- At presentation:1 collect blood for culture2 and serum for IgM and acute MAT
- At least 14 days and preferably not more than 28 days after acute MAT collection: collect serum for IgM and convalescent MAT3
For suspected leptospirosis cases presenting more than 10 days from symptom onset:
- At presentation:1 collect serum for IgM and acute MAT
- At least 14 days after acute MAT collection: collect serum for IgM and convalescent MAT3
1. Collect serum at presentation for PCR and IgM/MAT, preferably on the same day as treatment commences.
2. Culture is the gold standard for detection during the leptospiraemic phase and has greater sensitivity than PCR but takes time due to the six-week culture period used. Contact your local microbiology/pathology laboratory for further information on culture collection options.
3. Convalescent MAT testing and culture supports identification of infecting serovars which is of particular importance for public health surveillance and outbreak detection.
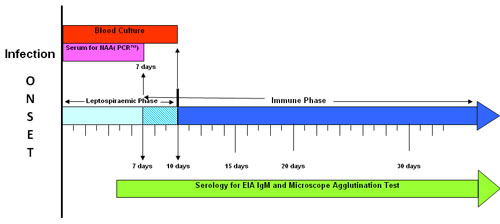
Source: WHO/FAO/OIE Collaborating Centre for Reference and Research on Leptospirosis. National leptospirosis surveillance report no 18. Queensland Health Forensic and Scientific Services: Coopers Plains, 2009.
Objects of surveillance
To monitor the epidemiology of leptospirosis in Queensland and inform public health initiatives.
Public Health Significance and Occurrence
Leptospirosis occurs worldwide (except the polar regions), but is most common in tropical and subtropical areas, and is common in Queensland. (3,4)
Leptospirosis is predominantly a disease reported in males, linked to occupation. Occupations reported as high risk include those in agriculture, such as the banana industry, meatworkers and dairy farmers. (4,5) The disease is also a recreational hazard for bathers, campers and sportspeople exposed to contaminated waters. (3,6) There have been several cases reported among white water rafters in Queensland. (7) Outbreaks can occur among those exposed to river, stream, canal and lake water contaminated by the urine and tissues of infected domestic and wild animals. Clusters have been associated with domestic and workplace rodent infestations. (8)
High rainfall and flooding likely contributes to the survival, growth and environmental contamination of Leptosira in water and soil and is a major risk factor for transmission. (4,9,10) Leptospirosis appears to be increasing globally due to the combined effects of climate change, flooding, urban population growth and agricultural intensification. (3,11)
Notifications of leptospirosis in Queensland
During the period between 2019–2023, there were on average 99 cases of leptospirosis notified annually in Queensland. The notification rate per year was fourfold greater in males than in females, and in most years approximately twofold greater in Aboriginal and Torres Strait Islander Queenslanders. Approximately half of notified cases each year occur in Cairns and Hinterland Hospital and Health Service.
Notifications of leptospirosis in Queensland: 2019-2023
| Year | Number of notifications | *Notification rate (per 100,000 population per year)# |
2019 | 56 | 1.1 |
2020 | 82 | 1.6 |
2021 | 155 | 3.0 |
2022 | 118 | 2.2 |
2023 | 86 | 1.6 |
* Number of notifications especially in most recent periods are subject to change due to the dynamic nature of the surveillance system.
# Notification rates were calculated based on Estimated Resident Population
Clinical Features
Leptospirosis is a zoonotic bacterial disease with varying manifestations. It is usually self-limiting and often clinically unapparent. Clinical illness lasts from a few days to three weeks or longer. Generally, there are 2 phases in the illness: the leptospiraemic or acute, phase during the first 7–14 days, followed by the convalescent or immune phase. Recovery in untreated cases can take several months. The early phase of the illness is characterised by fever with sudden onset, headache, chills, and severe myalgia (calves and thighs). Other symptoms may include nausea, vomiting, abdominal pain, diarrhoea, cough, photophobia, and rash. Conjunctival suffusion, the pathognomonic finding of leptospirosis, occurs in about 30% of cases. (3,12,13)
Between five to 10% of clinical cases progress to severe late-phase manifestations. These include:
- prolonged fever
- jaundice
- renal failure
- bleeding
- respiratory insufficiency with or without haemoptysis
- hypotension
- myocarditis
- aseptic meningitis
- mental confusion
- depression. (3)
Case fatality rate of severe leptospirosis is estimated between 5–50% and is associated with older age, and multiple comorbidities. Acute respiratory distress syndrome, pulmonary hemorrhage, renal impairment and hypotension are manifestations associated with poor outcomes. (3,14)
Cases are often under-recognised or misdiagnosed as dengue, malaria and influenza. (3)
Reservoir
Pathogenic Leptospira colonise the renal tubules of wild and domestic animals and can survive in the environment for weeks. (11) Infected animals may excrete leptospires intermittently or persistently for months, years, or their lifetime. (3) Leptospira have been isolated from virtually all mammalian species. Cattle, pigs, and dogs serve as major reservoir hosts.
Serovars generally vary with the animal affected. Rats are a common source for a number of serovars, the main serovars detected in Australia associated with rats and rattus species are Arborea, Zanoni and Australis. Refer to appendix A for further information on serovars and animal hosts.
Mode of Transmission
Transmission can be via (3,12):
- Contact of the skin (especially if abraded) or mucous membranes with moist soil, vegetation (sugar cane and banana farms have been implicated) or water that has been contaminated with the urine of infected animals.
- Direct contact with urine or tissues of infected animals including rodents.
- Ingestion of food or water that has been contaminated with urine of infected animals.
- Inhalation of droplet aerosols of contaminated fluids
- Contact with contaminated water during outdoor activities such as swimming, wading and white-water rafting in rivers, lakes and dams.
While very rare, human to human transmission has been reported through sexual contact or breastfeeding.
Incubation Period
Usually 5-14 days, with a range of 2-30 days. (3)
Period of Communicability
Leptospires may be excreted in the urine, usually for one month, although prolonged excretion has been observed in humans and in animals for months, even years, after acute illness. (4,12)
Susceptibility
Males and young adults are generally at higher risk for the development of symptomatic disease. Following infection, immunity is likely for the specific serovar if antibody titres are sufficient, but may not protect against infection with another serovar. (3,12)
A vaccine to prevent human disease is not available in Australia. (15)
Management
Cases
Investigation
Public Health follow up is recommended for all cases within 5 working days of notification, with further investigations guided by the laboratory and clinical features of the case.
See Laboratory Aspects section above for advice on specimen collection and timing.
The principles of investigation are:
- Confirm the diagnosis. When notified on the basis of a single MAT, contact the treating medical practitioner for clinical information and attempt to obtain a second sample for further serological testing. When notified on the basis of a positive enzyme-linked immunosorbent assay IgM result, await MAT testing result. Where a case is confirmed by nucleic acid testing, a MAT test is required to identify the serovar.
- Identify likely source of infection. For confirmed cases, in consultation with the treating medical practitioner, attempt to identify the source of infection such as exposure to urine or tissues of infected animals or contaminated water.
- Refer for further investigation if appropriate. If occupational exposure is suspected, obtain patient consent to notify Workplace Health and Safety Queensland.
Restriction
Nil; use standard precautions in the clinical setting.
Counselling
The case should be advised of the nature of the infection and its mode of transmission.
Treatment
Treatment is the responsibility of the treating clinician. Treatment is most effective when given early in the illness and according to recommendations in the current edition of Therapeutic Guidelines. Doxycycline is the preferred antibiotic for empirical therapy because it also treats rickettsial infections that present similarly to leptospirosis.
Contacts
Contact Tracing
Contact tracing may be particularly pertinent for recreational events and other potential common exposures. Examples could include rafting with a confirmed case, swimming or fishing in freshwater creeks, rivers or other water sources.
Definition
Any person exposed to the likely source of infection for the case.
Management
Advise contacts to seek medical advice and testing if symptoms occur.
Prophylaxis
Nil for contacts of sporadic cases.
Counselling
Contacts should be advised of the nature of the infection and its mode of transmission, and to seek medical advice early if symptoms develop.
Community or workplace outbreaks
Investigation should include identifying a common source of exposure. Consideration for both potential community and occupational exposures is necessary. To mitigate identified sources, consider appropriate preventative measures to minimise infection risk and environmental contamination.
Community outbreaks of leptospirosis in Queensland have historically been associated with extreme weather events, such as floods and cyclones. The public health response focuses on awareness raising among health care providers, e.g. GP and ED alerts, community messaging concentrating on minimising exposure risks, e.g. promoting appropriate clothing and footwear during disaster response and recovery, and surveillance to ascertain cases and identify outbreaks.
Chemoprophylaxis with medications such as doxycycline may be considered in clusters in specific occupational settings. There is very limited evidence supporting antibiotic chemoprophylaxis for leptospirosis (16) and the logistical challenge of providing medication to large numbers of people during disasters renders community chemoprophylaxis unfeasible in most instances. (9)
Preventive Measures
A vaccine to prevent human leptospirosis is not available. Prevention measures should be targeted at reducing exposure to infected animals, and contaminated environments, targeting the individual, community, and the broader environment as well as animals.
Individual
- Promote public awareness of possible transmission risk when undertaking recreational water-based activities including wading, swimming, white water rafting in potentially contaminated waters especially after heavy rain and flooding.
- Provide staff working in hazardous occupations with appropriate protective equipment such as boots, gloves, and aprons.
- Covering wounds with a waterproof dressing may reduce risk of infection in people with possible occupational or recreational exposures such as working with animals or exposure to mud, soil or water that may be contaminated with animal urine.
- Encourage frequent hand washing, especially prior to ‘hand-to-mouth’ activities (eg. eating and smoking), or after handling animals.
Community, public health, and environmental health
- Identify and manage contaminated waters and soil such as by draining the waters.
- Where possible create physical barriers, such as closing open sewers.
- Minimise local flooding risk by ensuring that streams and drains are not blocked.
- Homes that have been contaminated by flood waters should be cleaned and disinfected (the leptospires are rapidly killed by disinfectants and desiccation).
- Warning signs should be placed in high-risk areas and the community and clinicians should be alerted about leptospirosis during high-risk times (such as flooding).
- It is important that sanitation and infrastructure is maintained and repaired as soon as possible.
Animals
- Control rodents in homes and recreational areas-reduce rodent infestation with regular rubbish removal and adequate sanitation.
- Segregate infected domestic animals.
- Maintain good hygiene practices (such as regular handwashing) while caring for or handling of animals (and avoid exposure to the urine or other bodily fluids from infected animals).
References
1. Queensland Health. What is leptospirosis [Internet]. corporateName=The State of Queensland; jurisdiction=Queensland; 2020 [cited 2023 Sep 25]. Available from: https://www.health.qld.gov.au/public-health/forensic-and-scientific-services/testing-analysis/disease-investigation-and-analysis/leptospirosis-reference-laboratory/what-is-leptospirosis
2. Australian Government Department of Health and Aged Care. Australian Government Department of Health and Aged Care. Australian Government Department of Health and Aged Care; 2004 [cited 2023 Sep 25]. Leptospirosis – Surveillance case definition. Available from: https://www.health.gov.au/resources/publications/leptospirosis-surveillance-case-definition?language=en
3. Lau C, Craig S. Leptospirosis. In: Control of Communicable Diseases Manual [Internet]. American Public Health Association; 2015 [cited 2023 Sep 25]. (Control of Communicable Diseases Manual). Available from: https://ccdm.aphapublications.org/doi/full/10.2105/CCDM.2745.090
4. Lau CL, Skelly C, Dohnt M, Smythe LD. The emergence of Leptospira borgpetersenii serovar Arborea in Queensland, Australia, 2001 to 2013. BMC Infect Dis. 2015 Jun 14;15(1):230.
5. Taunton C, Hayek CE, Field E, Rubenach S, Esmonde J, Smith S, et al. Undetected serovars: leptospirosis cases in the Cairns region during the 2021 wet season. Commun Dis Intell [Internet]. 2022 Nov 17 [cited 2023 Sep 25];46. Available from: https://www1.health.gov.au/internet/main/publishing.nsf/Content/2A15CD097063EF40CA2587CE008354F1/$File/undetected_serovars_leptospirosis_cases_in_the_cairns_region_during_the_2021_wet_season.pdf
6. Cunningham S, Marquardt T, Moodley N. Leptospirosis Cluster Following Recreational 4-Wheel Driving and Vehicle Recovery Exposure. Int J Infect Dis. 2022 Mar 1;116:S72–3.
7. Smythe L, Dohnt M, Symonds M, Barnett L, Moore M, Brookes D, et al. Review of leptospirosis notifications in Queensland and Australia: January 1998 – June 1999. 2000;24(6).
8. Katelaris AL, Glasgow K, Lawrence K, Corben P, Zheng A, Sumithra S, et al. Investigation and response to an outbreak of leptospirosis among raspberry workers in Australia, 2018. Zoonoses Public Health. 2020;67(1):35–43.
9. Smith JKG, Young MM, Wilson KL, Craig SB. Leptospirosis following a major flood in Central Queensland, Australia. Epidemiol Infect. 2013 Mar;141(3):585–90.
10. Wright H, Goot K, Rogers B. Spirochaetes and sunshine: leptospirosis in the aftermath of the Queensland floods. Med J Aust. 2012;196(8):500–2.
11. Lau CL, Townell N, Stephenson E, Van Den Berg D, Craig SB. Leptospirosis: An important zoonosis acquired through work, play and travel. Aust J Gen Pract. 2018 Mar 1;47(3):105–10.
12. World Health Organization. leptospirosis - Fact Sheet [Internet]. World Health Organization; 2009. Available from: https://www.who.int/publications/i/item/B4221
13. Rajapakse S. Leptospirosis: clinical aspects. Clin Med. 2022 Jan;22(1):14–7.
14. Smith S, Liu YH, Carter A, Kennedy BJ, Dermedgoglou A, Poulgrain SS, et al. Severe leptospirosis in tropical Australia: Optimising intensive care unit management to reduce mortality. PLoS Negl Trop Dis. 2019 Dec 2;13(12):e0007929.
15. Teixeira AF, Fernandes LGV, Cavenague MF, Takahashi MB, Santos JC, Passalia FJ, et al. Adjuvanted leptospiral vaccines: Challenges and future development of new leptospirosis vaccines. Vaccine. 2019 Jul;37(30):3961–73.
16. Win TZ, Perinpanathan T, Mukadi P, Smith C, Edwards T, Han SM, et al. Antibiotic prophylaxis for leptospirosis. Cochrane Database Syst Rev. 2024 Mar 14;3(3):CD014959.
Further reading
Win TZ, Perinpanathan T, Mukadi P, Smith C, Edwards T, Han SM, Maung HT, Brett-Major DM, Lee N. Antibiotic prophylaxis for leptospirosis. Cochrane Database of Systematic Reviews 2024, Issue 3. Art. No.: CD014959. DOI: 10.1002/14651858.CD014959.pub2. Accessed 27 March 2024.
Appendix A: Microscopic Agglutination Test (MAT) serovar panel and commonly recognised animal hosts
| Serovar | Serogroup | Commonly recognised animal host (list is not exhaustive) |
|---|---|---|
| Pomona | Pomona | Feral and domestic pigs, horses, deer, sheep |
| Hardjo | Sejroe | Cattle, sheep, platypus |
| Tarassovi | Tarassovi | Domestic pigs, |
| Grippotyphosa | Grippotyphosa | Native rodents such as Melomys (or mosaic-trailed rat), cattle |
| Celledoni | Celledoni | Rodents and bandicoots |
| Copenhageni | Icterohaemorrhagiae | Rattus norvegicus (brown rat) domestic dogs |
| Australia | Australis | Rattus species |
| Zanoni | Pyrogenes | Rattus species |
| Robinsoni | Pyrogenes | Rattus species |
| Canicola | Canicola | Rodents (also for serovars Bindjei & Broomi) |
| Kremastos | Hebdomadis | Rodents and bandicoots |
| Szwajizak | Mini | Rodents |
| Medanensis | Sejroe | Cattle ( not common) |
| Bulgarica | Autumnalis | Rattus species (not common) |
| Cynopteri | Cynopteri | Not Australian, found in bats in Indonesia |
| Arborea | Ballum | Introduced rodents (recorded in Australia since 1966) |
| Bataviae | Bataviae | Not Australian, found in rodents throughout Indonesia and Asia |
| Djasiman | Djasiman | Not Australian, found in rodents in Indonesia |
| Javanica | Javanica | Not Australian, found in rodents throughout Indonesia and Asia |
| Panama | Panama | Not Australian, common in Central and South America |
| Shermani | Shermani | Not Australian, some serogroup members in China |
| Topaz | Tarassovi | Macropods |